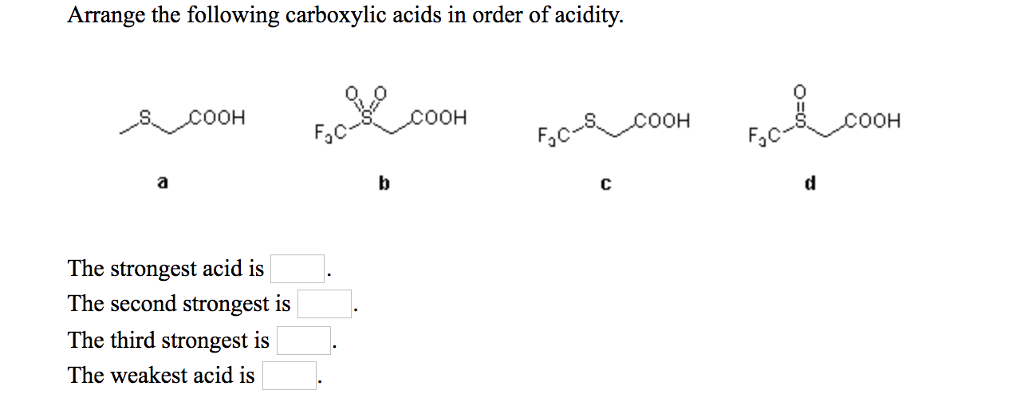
  They will be having Maximum authority due to the presence of two grooming groups and they will be showing negative. Oh the substitution that are linked to each compound, we can write the increasing order increasing order paul activity for the above compound as we will be having compound B. CICH,-COOH ICH,-COOH FCH, COOH CH, COOH The strongest acid is The. We have seen double bond here and always this is labeled as compounding now according to the according to the inductive effect. Solution for Review opics) Arrange the following carboxylic acids in order of acidity. This problem has been solved See the answer Arrange the following carboxylic acids in order of acidity. Acidity increase with the presence of a group with -I effect in the alkyl group. can read more about the inductive effect and the pKa of carboxylic acids in. As we have learnt, Acidic strength of carboxylic acid. There are two bromine atoms linked together. Example: Rank the following compounds in order of increasing acidity without. Science Chemistry Q&A Library Review opics) Arrange the following carboxylic acids in order of acidity. And moving on to the structure of the last some pounds On the first carbon. Electron withdrawing groups like -NO2, increases the acidity of. Therefore it increases electron density as a result removal of proton becomes difficidt. NO2CH2COOH is most acidic among the given three compounds. Incase of acetic acid, methyl group is electron doner. 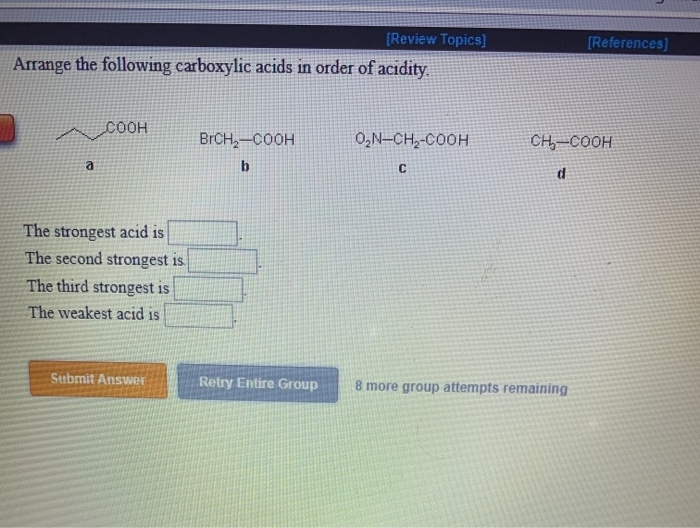 Chlorine is electronegative element, it attracts the electrons towards itself, as a result removal of proton from chloroacetic acid becomes easier. Double Bondo which and onto the veg bond here we have ch three group. Acetic acid < Fromic acid< Chloroacetic acidProton donors are acids. Moving on to the structure of the second compound that we have here we have double bond poet basically all the compounds are carb oxalic acid groups, compounds they contain the groups of delicacy. This is the structure of the first compound here we have a witch labor last compound and on the second carbon we have Sinai group linked towards the dash bond. The answer to the question above of B: e Amines (e) are the least acidic acids of the group drawn above: amide anions (R2N-) are the least stable of the conjugate. Similarly, factors which facilitate the breaking of the X-H bond will also increase the acidity of HA. As you should know, acidity stems from the ability of a compound to release protons in the medium, so acids are proton donors. 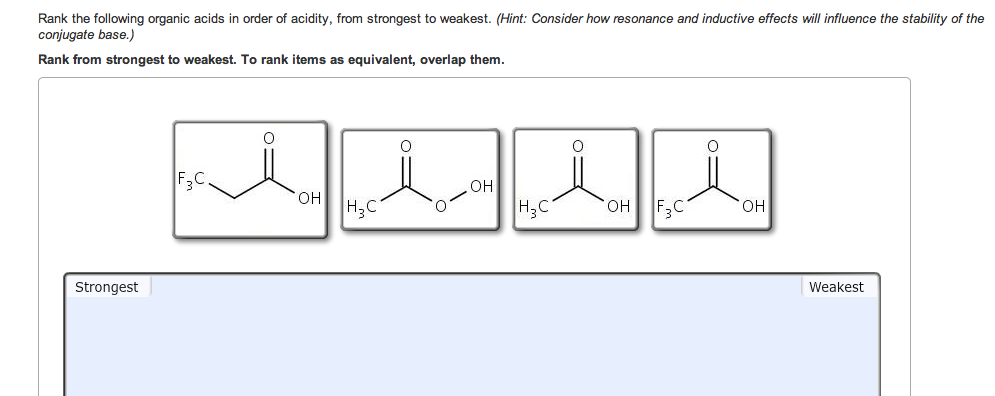 In this question we are given with views of compound for which we have to write the correct increasing order of authority of the compound. equilibrium of this process and result in a smaller pKa (i.e. The ranking of these acids in order of increasing acidity is: CH3CH2COOH 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
